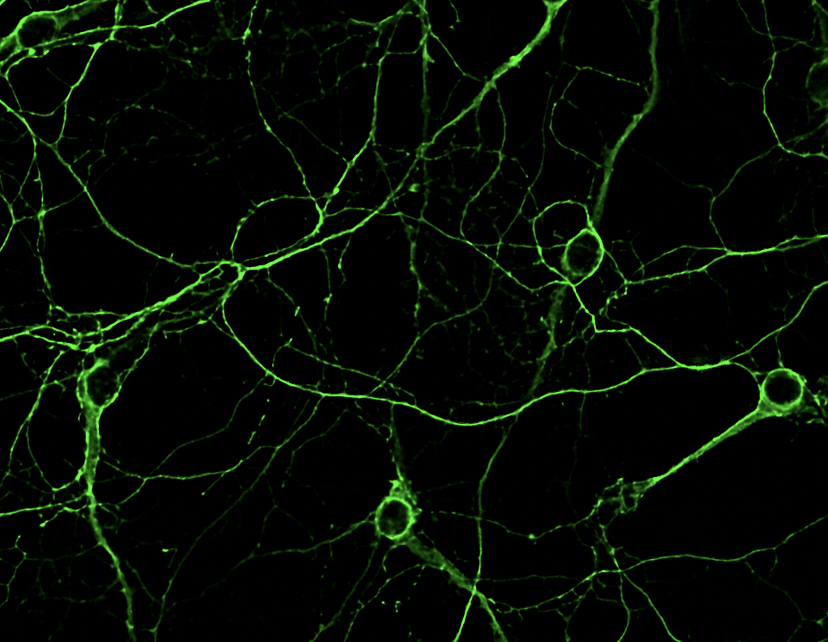
The Vossel Lab studies molecular mechanisms of tau through techniques including proximity ligation assay, live cell signaling assays, slice electrophysiology, and electroencephalography. These assays allow us to see which molecules of interest the tau protein interacts with and provides a detailed picture of the synaptic integrity and plasticity within brain regions of interest.
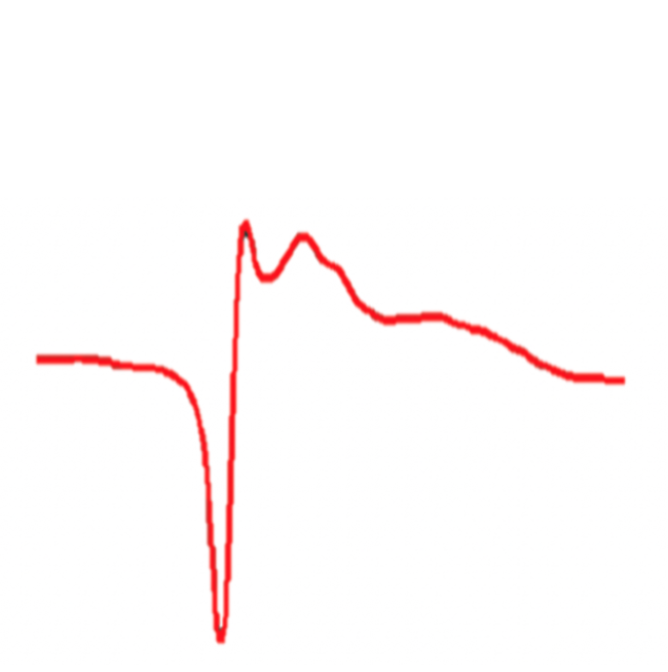
Dr. Vossel’s clinical studies with Alzheimer’s disease (AD) patients revealed that over 40% of patients exhibit silent epileptiform activity, mostly occuring during sleep, and this subclinical phenomenon can accelerate disease progression. Additionally, an estimated 20% of Alzheimer's patients develop seizures and associated cognitive decline. Therefore, up to 60% of Alzheimer's patients have seizures or epileptiform activity and could benefit from anti-seizure treatments. In a Phase 2a study, we showed that low doses of the anti-seizure drug levetiracetam improves spatial memory and executive function in patients with AD and detectable epileptiform activity. The Vossel lab has also demonstrated that patients with dementia with Lewy bodies have increased risk of seizures and myoclonus, which can hasten cognitive decline. To further understand seizure/epileptiform activity’s contribution to network dysfunction in dementia, the Vossel Lab uses electroencephalography (EEG) recordings in mice treated with antiseizure drugs or tau modifications. Studies using EEG recordings in models of neurodegenerative disease provide valuable insight into assessing how the blockade of certain biological processes may ameliorate overall network dysfunction.
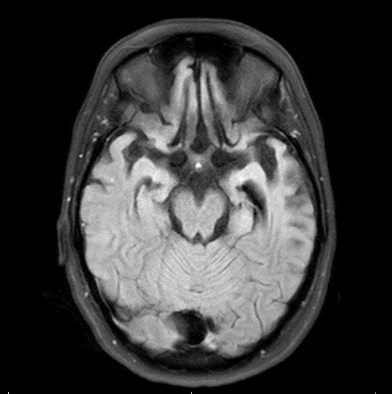
Frontotemporal dementia (FTD) rarely occurs in individuals under the age of 30, and genetic causes of early-onset FTD are largely unknown. Our lab discovered retrotransposition of the MATR3 gene, a DNA/RNA binding protein, associated with early-onset FTD. This is a novel mutation mechanism of MATR3, although other mutations in MATR3 have been associated with FTD/ALS pathology previously. Retrotransposons are not often implicated in neurodegenerative diseases; thus, the Vossel lab is clarifying the potential role of MATR3 retrotransposition in early-onset FTD.
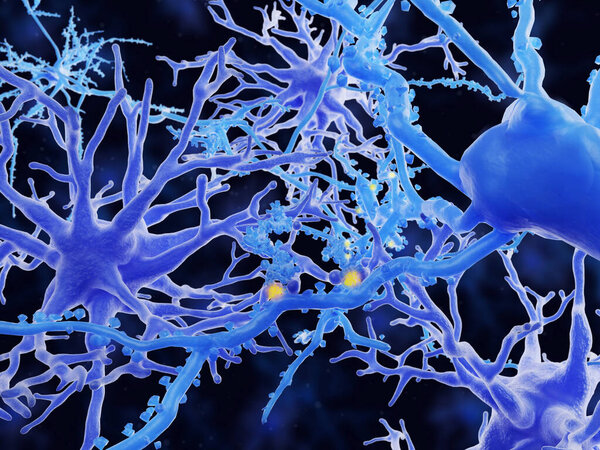
The Vossel lab is collaborating with Drs. Baljit Khakh and James Wohlschlegel to study the effects of aberrant network activity and neuroinflammation on protein expression in astrocytes and neurons in models of aging and Alzheimer's disease (AD). This study is funded by an NIH R01 award. We are using advanced proteomic methods to identify cell-specific proteomes in the cytosol and plasma membrane. We will test how these proteomes change in association with aberrant network activity, neuroinflammation, and cognitive decline in AD.
We have several ongoing clinical studies including studies initiated by Dr. Vossel and investigations that are part of nationwide consortia as follows:

The Vossel lab is collaborating with UCLA Epilepsy Program, Neurosurgery, and Neuropathology to study the relationship between seizures and cognitive decline in mesial temporal lobe epilepsy (MTLE) and Alzheimer's diseaes (AD). We received an NIH R01 award to carry out the following goals: 1) In patients with AD and mild cognitive impairment (MCI) who exhibit epileptiform activity and/or pathological high frequency oscillations (pHFOs) on overnight EEG, perform hippocampal depth electrode recordings over 5 days to assess for hippocampal seizures and further characterize the hippocampal neurophysiology. Participants will be given brivaracetam to suppress epileptic activity and maintained on the most effective dose of brivaracetam for a year in an open-label clinical trial; 2) Carry out pathological analysis of hippocampal specimens for AD pathology from participants with MTLE who undergo surgical resection and follow these patients longitudinally; 3) In a rat model of AD with epilepsy, perform electrophysiological and behavioral testing before and after treatment with brivaracetam. EEG findings will be compared with data obtained from the intrahippocampal kainate rat model. (Site PIs: Keith Vossel, MD, MSc, Jerome Engel, MD, PhD, Richard Staba, PhD)
Contact: Aisha Mohammed at (310) 825-2600 or amohammed@mednet.ucla.edu,
or Sydney Kilgore at (310) 825-3198 or Skilgore@mednet.ucla.edu to participate in this study or for more information.
BIND is the main observational study supporting various research projects within the Easton Center. The purpose of BIND is to collect a large amount of information from tests of brain function and cognitive performance over a period of time to help researchers learn more about and develop new therapies for neurodegenerative diseases. This study seeks to enroll participants willing to undergo cognitive testing, brain imaging (PET and MRI), blood draws, optional lumbar puncture, and an overnight sleep study or 2-hour EEG. Eligible participants may have normal cognition or be diagnosed with mild cognitive impairment, Alzheimer’s disease, dementia with Lewy bodies, Parkinson’s disease with dementia, frontotemporal dementia, primary progressive aphasia, progressive supranuclear palsy, corticobasal degeneration, transient epileptic amnesia, or adult-onset epilepsy. Participants will be evaluated for as long as they are willing and able to participate. (Site PI: Keith Vossel, MD, MSc)
Contact: Aisha Mohammed at (310) 825-2600 or amohammed@mednet.ucla.edu,
or Sydney Kilgore at (310) 825-3198 or Skilgore@mednet.ucla.edu to participate in this study or for more information.
NIA-AD FBS is a longitudinal observational study recruiting families with multiple members diagnosed with Alzheimer’s disease (AD) or related disorders for genetic research. The study seeks to enroll families with 3+ individuals willing to participate. For families to be included, at least one sibling must have an established diagnosis of AD or have a diagnosis of a related disorder such as frontotemporal dementia or dementia with Lewy bodies. The family will also require an informant. Those who choose to participate in this study will undergo cognitive testing, MRIs, and blood draws every 18 months for up to 5 years. (Site PI: Keith Vossel, MD, MSc)
Contact: Aisha Mohammed at (310) 825-2600 or amohammed@mednet.ucla.edu,
or Sydney Kilgore at (310) 825-3198 or Skilgore@mednet.ucla.edu to participate in this study or for more information.
MarkVCID aims to identify and validate biomarkers for the small vessel diseases of the brain that produce vascular contributions to cognitive impairment and dementia (VCID). As a site for the larger NIH-funded MarkVCID consortium, UCLA’s goal is to deliver high-quality biomarkers ready for use in clinical trials aimed at generating scientific breakthroughs in the understanding and treatment of VCID. This study seeks to enroll participants willing to undergo cognitive testing, MRI, and a blood draw. Eligible participants will either be normal controls, have a diagnosis of mild cognitive impairment, or have a diagnosis of mild dementia. (Site PIs: Jason Hinman, MD and Keith Vossel, MD, MSc)
Contact: Marissa Thirion at (310) 206-5178 or mthirion@mednet.ucla.edu to participate in this study or for more information.